血糖试剂条印刷电极是血糖检测的核心组件之一,其原理涉及电化学检测技术与生物酶催化反应的结合,通过将电极按照设计图纸印刷在基底材料上,实现对血液中葡萄糖浓度的快速、准确测量。以下从结构、工作原理及关键技术细节展开说明:
血糖试剂条的印刷电极通常由基底材料和多层印刷电极图案组成,核心结构包括:
1. 基底材料
多为绝缘性良好的高分子材料(如聚氯乙烯PVC、聚酯PET等),起到支撑电极和绝缘的作用。
2. 电极系统
通过丝网印刷、喷墨印刷等技术,将导电材料(如碳浆、银/氯化银浆)印刷在基底上,形成三个关键电极:
1) 工作电极(Working Electrode,WE):发生葡萄糖氧化反应的核心区域,表面通常固定有葡萄糖氧化酶(GOD)或葡萄糖脱氢酶(GDH)等生物酶。
2) 参比电极(Reference Electrode,RE):提供稳定的基准电势,确保工作电极电势的准确性(常用银/氯化银Ag/AgCl)。
3) 对电极(Counter Electrode,CE):与工作电极形成电流回路,平衡电子转移,避免参比电极被极化。
3. 试剂层
覆盖在工作电极表面,包含生物酶(催化葡萄糖反应)、电子媒介体(如铁氰化钾、二茂铁等,传递电子)及缓冲液(维持反应环境pH稳定)。
当血液样本滴加到试剂条上时,通过以下步骤完成血糖浓度检测:
血液中的葡萄糖扩散至试剂层,在生物酶的催化下发生氧化反应:
以葡萄糖氧化酶(GOD)为例:
葡萄糖+O₂→葡萄糖酸+H₂O₂(在GOD催化下)
若使用葡萄糖脱氢酶(GDH),则反应不依赖氧气,直接将葡萄糖氧化为葡萄糖酸内酯,同时氧化辅酶(如NAD⁺→NADH)。
氧化反应产生的电子需要通过电子媒介体传递到工作电极(避免直接与氧气反应导致的误差):
· 例如,H2O2在工作电极表面被氧化为O₂,释放电子(H2O2→2H⁺+O₂+2e⁻);
· 电子媒介体(如铁氰化钾[Fe(CN)₆]³⁻)接受电子后被还原为[Fe(CN)₆]4⁻,随后在工作电极表面重新被氧化为[Fe(CN)₆]³⁻,同时释放电子到电极上,形成电流。
工作电极与对电极之间施加恒定电压(由仪器控制,通常0.2~0.5V),电子转移产生的氧化电流强度与葡萄糖浓度成正比(遵循法拉第定律):
仪器检测到电流信号后,通过内部校准曲线将电流值转换为葡萄糖浓度(mmol/L或mg/dL),并显示结果。
印刷技术(如丝网印刷)的核心优势在于:
1. 低成本、大规模生产:通过模板快速将电极图案复制到基底上,适合批量制造。
2. 微型化与集成化:电极尺寸可精确控制在微米级,减少样本用量(仅需1~5μL血液)。
3. 稳定性提升:印刷的电极材料(如碳浆)与试剂层结合紧密,确保电子传递效率稳定。
· 快速检测:整个反应与检测过程可在10秒~30秒内完成。
· 高特异性:生物酶仅对葡萄糖有催化作用,减少其他物质干扰。
· 便携性:配合便携式血糖仪,适合家庭或即时检测(POCT)场景。
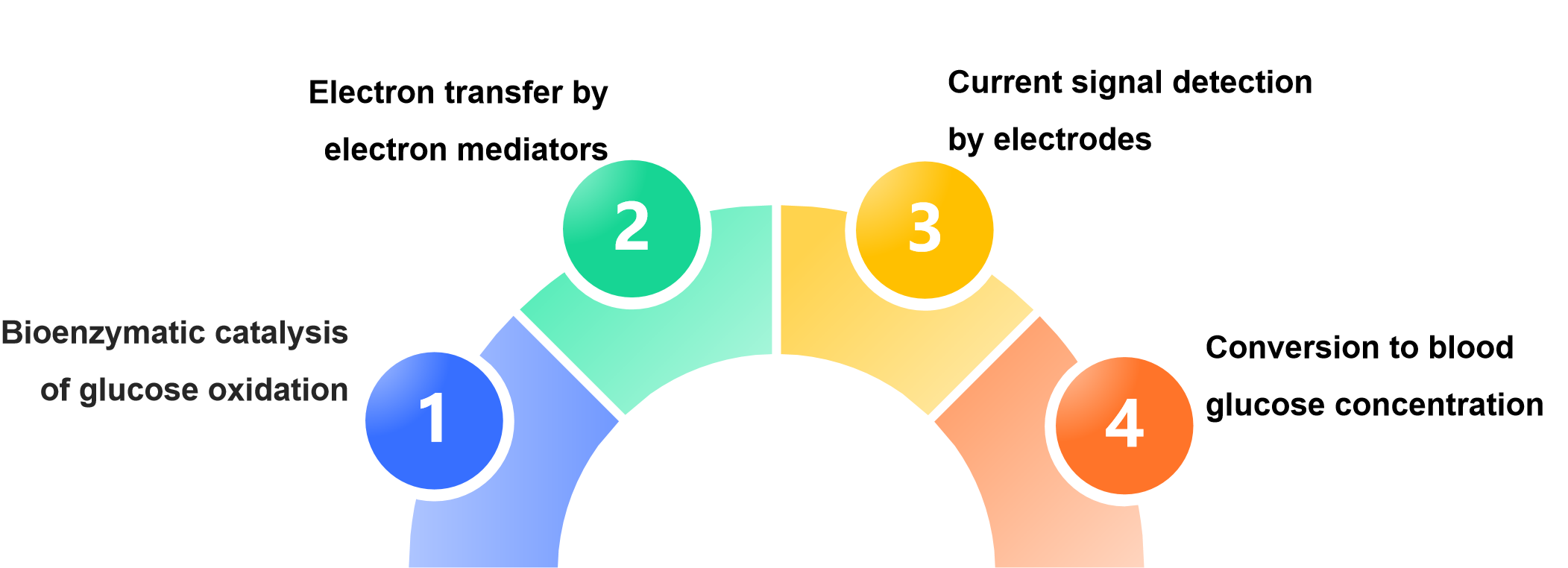
血糖试剂条印刷电极的原理是:
“生物酶催化葡萄糖氧化→电子媒介体传递电子→电极检测电流信号→转换为血糖浓度”的电化学过程,而印刷工艺则为这一过程提供了高效、低成本的微型化电极载体,是现代血糖检测技术普及的关键基础
The principle of printed electrodes in blood glucose test strips
A blood glucose test strip's printed electrode typically consists of a substrate material and multi-layer printed electrode patterns, with the core structure including:
1. Substrate material
Mostly high molecular materials with good insulation (such as polyvinyl chloride PVC, polyester PET, etc.), which serve to support the electrodes and provide insulation.
2. Electrode system
Conductive materials (such as carbon paste, silver/silver chloride paste) are printed on the substrate through screen printing, inkjet printing and other technologies to form three key electrodes:
1. Working Electrode (WE):The core area where glucose oxidation reactions occur, with bioenzymes such as glucose oxidase (GOD) or glucose dehydrogenase (GDH) usually immobilized on its surface.
2. Reference Electrode (RE):Provides a stable reference potential to ensure the accuracy of the working electrode's potential (silver/silver chloride Ag/AgCl is commonly used).
3. Counter Electrode (CE):Forms a current loop with the working electrode to balance electron transfer and prevent polarization of the reference electrode.
3. Reagent layer
Covering the surface of the working electrode, it contains bioenzymes (catalyzing glucose reactions), electron mediators (such as potassium ferricyanide, ferrocene, etc., for electron transfer), and buffer solutions (maintaining stable pH in the reaction environment).
When a blood sample is dropped onto the test strip, the blood glucose concentration is measured through the following steps:
Glucose in the blood diffuses into the reagent layer and undergoes oxidation reaction under the catalysis of bioenzymes:
· Taking glucose oxidase (GOD) as an example:
Glucose + O₂ → Gluconic acid + H₂O₂ (catalyzed by GOD)
· If glucose dehydrogenase (GDH) is used, the reaction does not depend on oxygen, directly oxidizing glucose to gluconolactone while oxidizing coenzymes (such as NAD⁺→NADH).
The electrons generated by the oxidation reaction need to be transferred to the working electrode through electron mediators (to avoid errors caused by direct reaction with oxygen):
· For example, H₂O₂ is oxidized to O₂ on the surface of the working electrode, releasing electrons (H₂O₂ → 2H⁺ + O₂ + 2e⁻);
· Electron mediators (such as potassium ferricyanide [Fe(CN)₆]³⁻) accept electrons and are reduced to [Fe(CN)₆]⁴⁻, which is then re-oxidized to [Fe(CN)₆]³⁻ on the surface of the working electrode, releasing electrons to the electrode to form a current.
A constant voltage (controlled by the instrument, usually 0.2~0.5V) is applied between the working electrode and the counter electrode. The oxidation current intensity generated by electron transfer is proportional to the glucose concentration (following Faraday's law):
· After detecting the current signal, the instrument converts the current value into glucose concentration (mmol/L or mg/dL) through an internal calibration curve and displays the result.
The core advantages of printing technologies (such as screen printing) are:
1. Low cost and mass production: The electrode pattern can be quickly copied onto the substrate through templates, suitable for mass manufacturing.
2. Miniaturization and integration: The electrode size can be precisely controlled at the micrometer level, reducing sample usage (only 1~5μL of blood is needed).
3. Improved stability: The printed electrode materials (such as carbon paste) are tightly combined with the reagent layer, ensuring stable electron transfer efficiency.
· Rapid detection: The entire reaction and detection process can be completed within 10~30 seconds.
· High specificity: Bioenzymes only catalyze reactions with glucose, reducing interference from other substances.
· Portability: Compatible with portable blood glucose meters, suitable for home or point-of-care testing (POCT) scenarios.
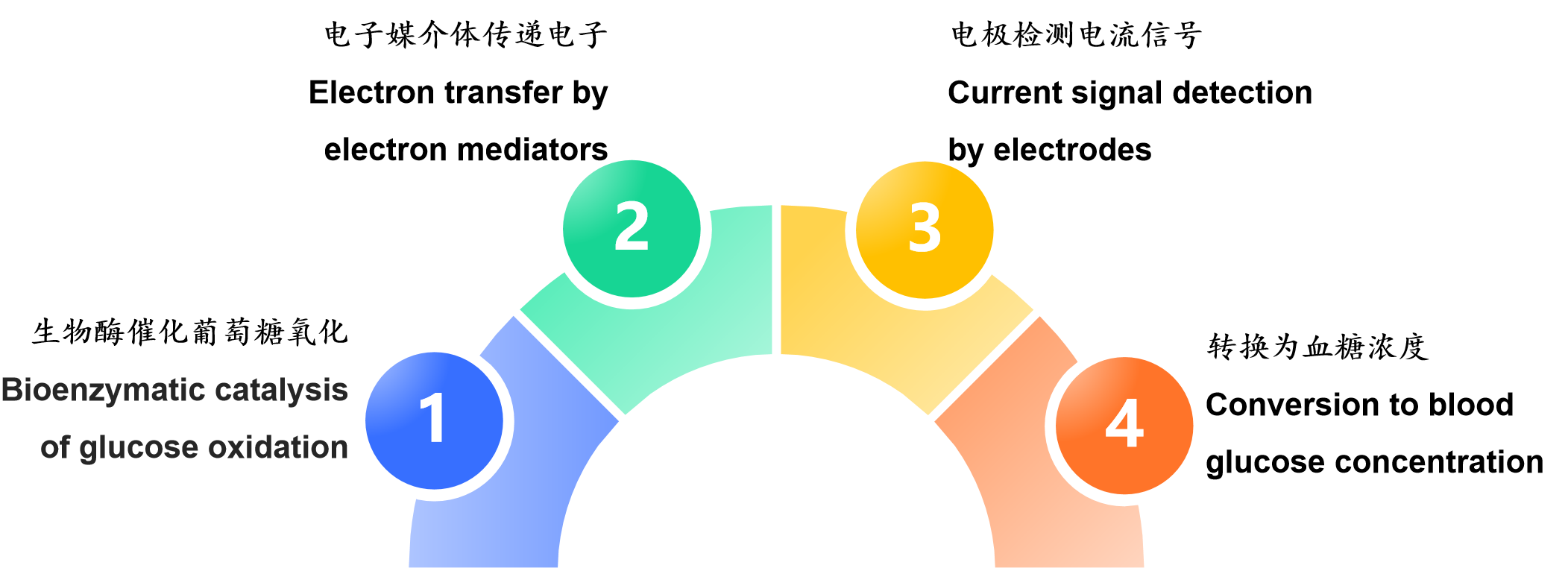
The principle of printed electrodes in blood glucose test strips is an electrochemical process of "bioenzymatic catalysis of glucose oxidation → electron transfer by electron mediators → electrode detection of current signals → conversion to blood glucose concentration". The printing process provides an efficient and low-cost miniaturized electrode carrier for this process, which is the key foundation for the popularization of modern blood glucose detection technology.